DHS H1N1 Weekly Situation Report
- 12 pages
- For Official Use Only
- October 19, 2009
Overview:
• As of 16 October, the number of Influenza-Like Illness (ILI) deaths in the U.S. was 537 this week compared to 405 deaths reported last week. (Source: HHS/CDC)
• As of 16 October, the most significant impacts of A(H1N1) remain increased ILI outpatient visits and influenza associated pediatric deaths. (Source: HHS/CDC)
Major Highlights:
• CDC provided updated interim guidance on the use of face masks and N95 respirators for decreasing exposure to A(H1N1). (Source: HHS/CDC)
• At least four of the ten HHS Regions are reporting N95 mask shortages. N95 respirator availability is extremely limited for new customers. Distributors are employing allocation strategies to prioritize existing customers based on average historical purchase. (Source: HHS/CDC)New Emergency Declarations:
• On 14 October, Illinois Governor Pat Quinn declared a public health emergency in the State of Illinois. (Source: DHS)
New Requests for Federal Assistance:
• None reportedCoordination Elements:
• RCTs (Regional Coordination Teams): All five RCTs partially activated (operating at 15 days per month). Conducting coordination. (Source: DHS)
• ESFs (Emergency Support Functions): ESF 8 working group is conducting twice weekly conference calls to update ongoing A(H1N1) response actions. (Source: HHS)
• FEMA H1N1 IMAT A (Incident Management Assistance Teams): All IMATs, including the National IMAT- East Team, completed A(H1N1) training. (Source: DHS/FEMA)…
Region A (Source Region A Team Leader):
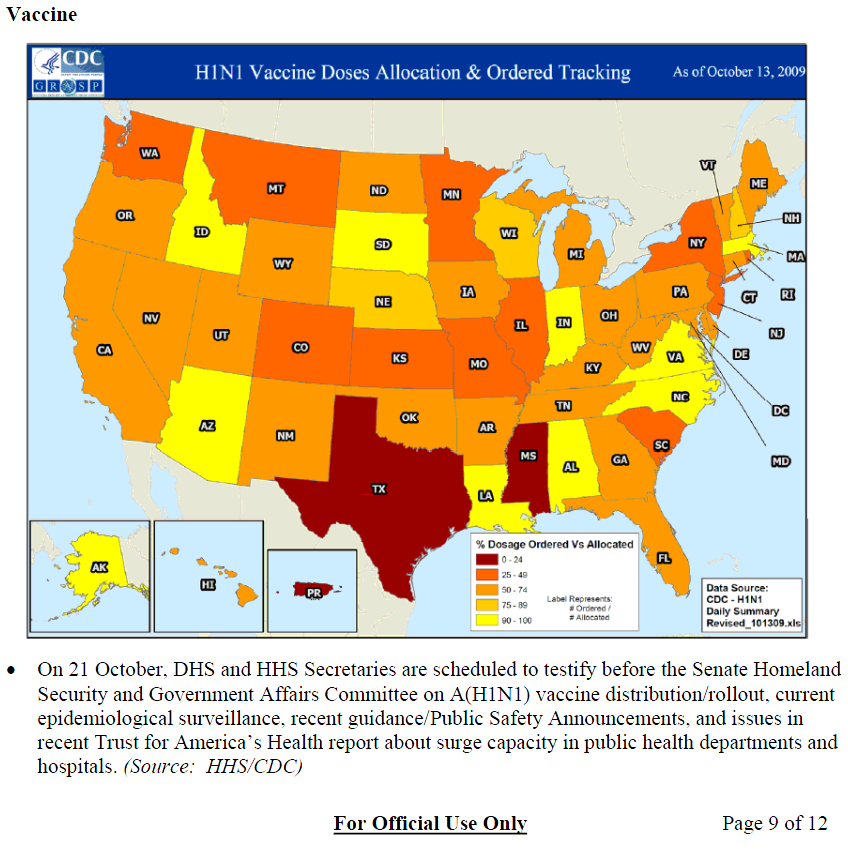
• State health officials have reported issues related to receiving significantly fewer doses of A(H1N1) vaccine than expected and the delayed distribution of seasonal flu vaccine injections. As a result, states have had to restrict and scale back distribution of A(H1N1) vaccines.Region B (Source Region B Team Leader):
• Wake County, North Carolina health officials closed four clinics after four days of immunizations due to depleted A(H1N1) nasal spray vaccine. Officials said they would reopen the clinics as soon as the vaccine is replenished.
• During HHS Region III’s conference call, State health departments reported continuing difficulties in obtaining Personal Protective Equipment (PPE), particularly N95 masks, from commercial suppliers and they may request PPE from the Strategic National Stockpile if shortages continue. When and if PPE becomes a true shortfall, requests will be coordinated through HHS.Region C (Source Region C Team Leader):
• On 16 October, Minnesota Department of Health hosted a closed meeting with HHS and CDC on Points of Dispensing for identified mission critical federal employees.
• In Saint Paul, Minnesota, Children’s Hospitals and Clinics of Minnesota recorded its second-highest patient volume ever at its Saint Paul Campus. Nearly half of the 195 emergency room patients were suffering from influenza-like illnesses.
• Sporadic release of both seasonal and A(H1N1) vaccine is providing a variety of challenges for state health departments and has resulted in cancellation of seasonal vaccine clinics.
• Region C remains currently unsupported for a Department of Defense Regional Joint Task Force (RJTF). The decision for appointment of the RJTF is presently with the Joint Staff and Secretary of Defense.Region D (Source Region D Team Leader):
• Barnes Jewish Children’s Hospital in St. Louis, Missouri reported opening two “reception/triage” tents in the hospital’s parking garage to take care of a very large overflow of patients with Influenza-Like-Illness.
• Many hospitals in CDC Region VI reported a high demand for services in emergency rooms.
• States have expressed concerns about visibility on delivery, timeliness, and amounts of A(H1N1) vaccines. CDC Region-VI DHHS/ASPR/OPEO has advised states to alert them regarding any shortfalls of PPE, vaccine, or antivirals.Region E (Source Region E Team Leader):
• HHS continues to administer grant programs to fund implementation of state and territory vaccination plans
• Federal agencies are conducting aggressive campaigns to promote Federal employee health, including vaccinations for seasonal flu and A(H1N1)
• FEMA Region X Telework Training was completed the week of 05 October
• American Samoa has not ordered its A(H1N1) vaccine due to cold chain problems
• Their vaccine storage facility does not have a backup generator and the facility has lost power several times during the past few weeks
• The American Samoa Governor’s office is working on the cold storage issue using on-island resources
• The Outer Pacific is experiencing several limiting factors including;
• A(H1N1) vaccine cold-storage and transportation (being addressed through HHS funding mechanisms)
• Limited laboratory presence which complicates surveillance efforts
• Limited trained personnel to administer vaccine in remote areas
• Real-time collaboration (being addressed by establishment of a page on the Virtual Joint Planning Office web site)…