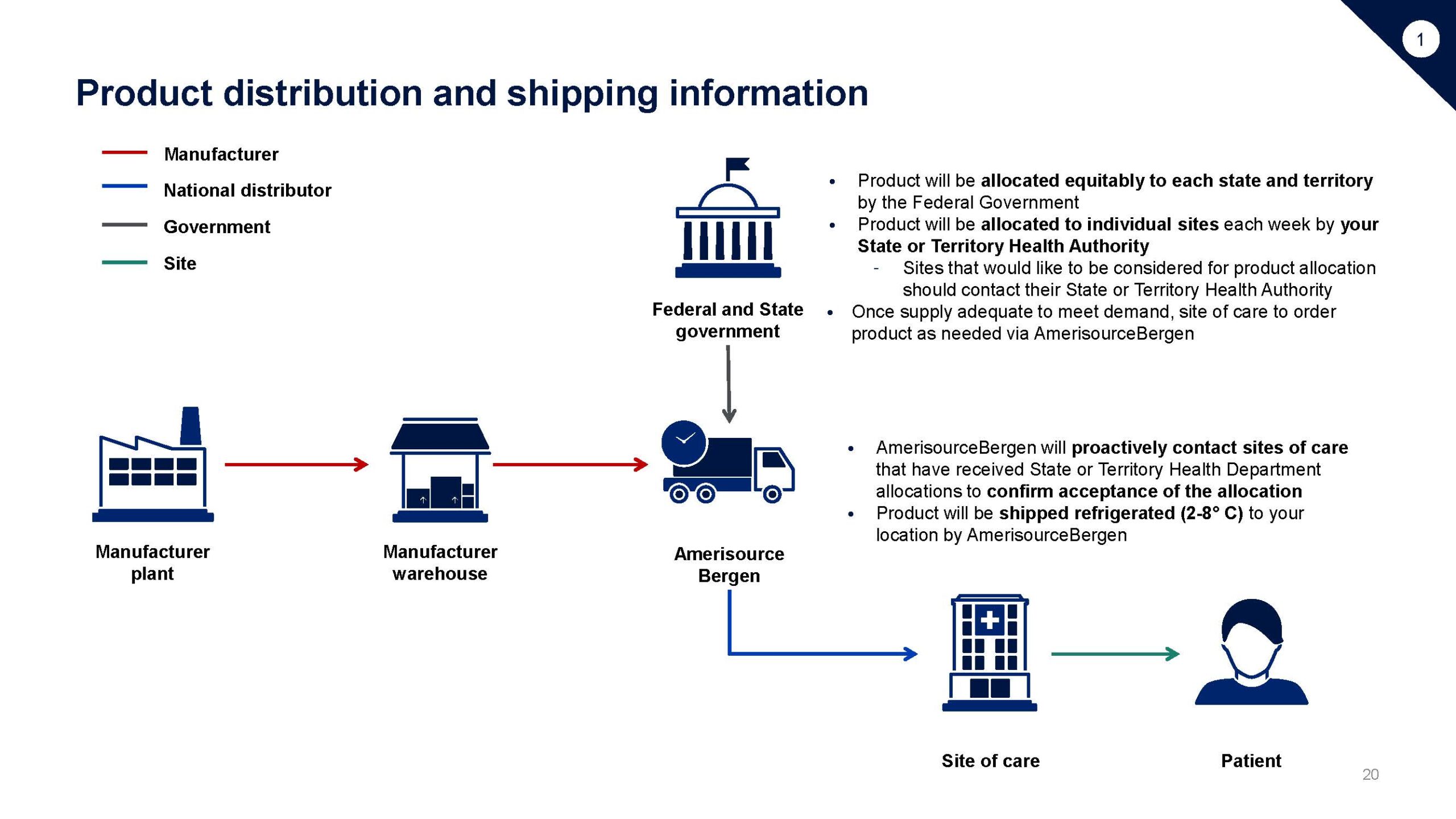
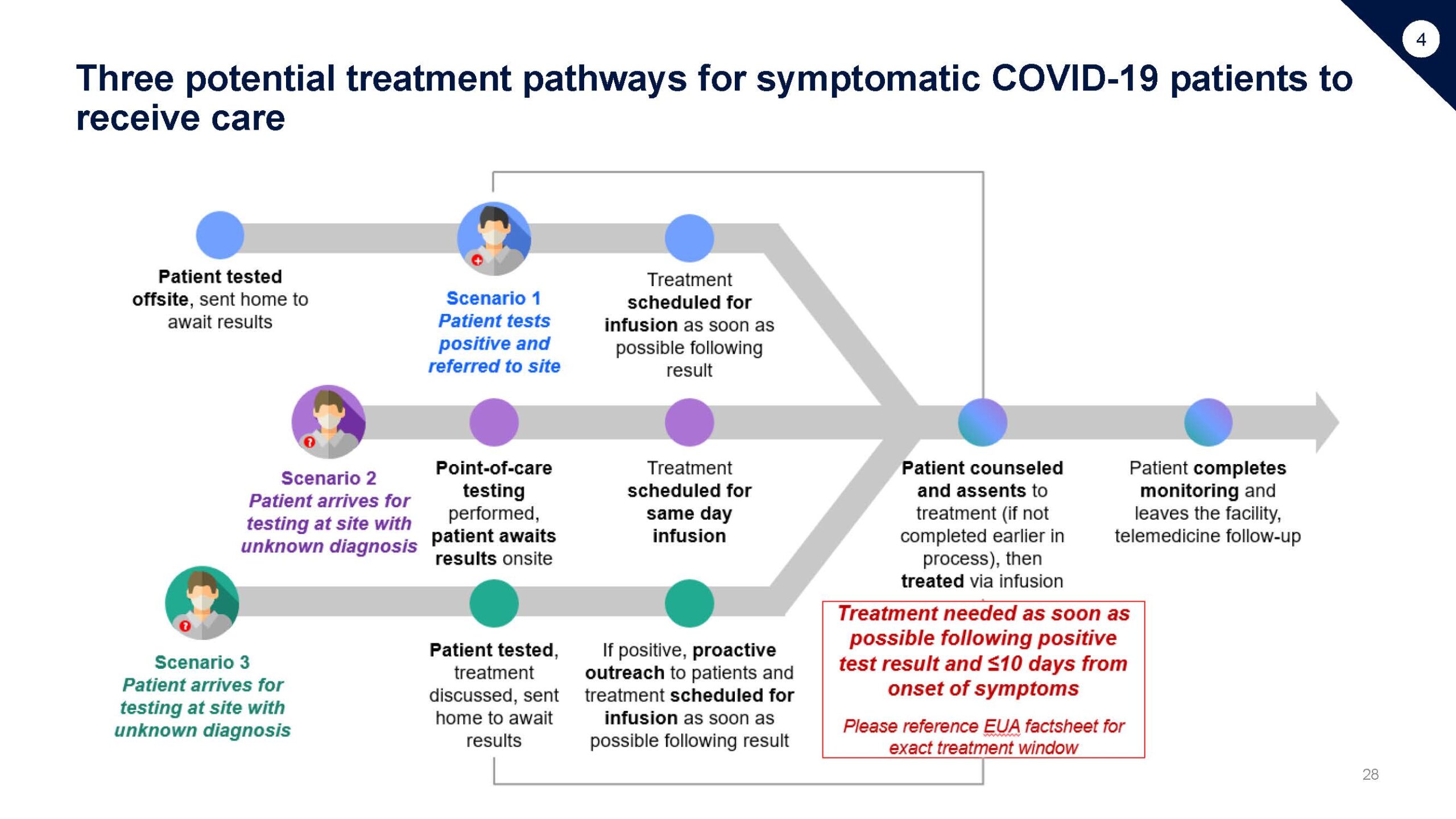
This playbook is intended to support sites interested in administering COVID-19 treatment under EUA including:
• Existing hospital or community-based infusion centers
• Existing clinical space (e.g. urgent care, emergency depts)
• Ad hoc new infusion sites (e.g. “hospitals without walls”)
• Long-term care facilities or home infusions with infusion delivery capability
Initial version of playbook focused on:
• Monoclonal antibody treatment
• Delivery via infusion
• Outpatient setting
This playbook will continue to evolve as other treatments and administration methods become available. We hope this playbook will be used to help healthcare facilities to implement monoclonal antibody treatment in an outpatient setting for those with COVID-19.
…